The studies on removal of vanadium were conducted using coconut shell. Adsorption efficiency has been evaluated. The effect of pH, contact time, adsorbent dose. concentration of metal, particle size and temperature were studied. The results reveals that Langmuir and Freundlich isotherms are followed during adsorption process. Thermodynamics parameter indicate the feasibility of the process. Kinetic studies have been performed to understand the mechanism of adsorption. Column studies have been carried out to compare these with batch capacities.
Keywords |
| Pentavalent Vanadium, Adsorption, Coconut Shell. |
INTRODUCTION |
| The Twentieth century started with an extensive damage to the natural resources
(Tomer, 1999). Unplanned industrilization, urbanization, pollution
explosion, change in lifestyle,
over exploitation of natural resources, commercial
establishment and modern
agricultural practices have degraded the
quality of environment. The main effects
being faced are : |
| • Continental invasion of air and water. |
| • Marine pollution through waste discharges. |
| • Release of variety of chemical and biological contaminants into the water
bodies, on land and in air. |
| • Ground water pollution. |
| • Acid rains and nuclear fallout. |
| These effects are not only covering the pollution of environment but also
are responsible
in creating genetic erosion in plants, animals including human
beings and microorganisms. Water is a prime natural resource and is a basic
human need. The availability of adequate water supply in terms of its quality and quality is essential
for the existence of life. |
| Water is available in nature as surface water and ground water through
the selfpurification mechanisms .like physical, chemical and microbiological
processes at natural bodies, are carried out in nature. However, natural water
is rarely suitable
for direct consumption to human beings. Rapid industrialization
and population
growth resulted to generation of large quantities of
wastewater and causing problem of their disposal. Industrial waste constitutes
the major source of various kinds of metal pollution fn natural water.
The presence of heavy metals in the enviroment has been of great concern
because of their increased discharge, toxic nature and other adverse effects
on the receiving streams. When the concentration of toxic metal ions exceed
tolerance limit, they may become real health concern (Singh and Lal, 1992).
There is an immediate need to introduce cleaner technologies to minimise
the pollution and to protect the degarding encironment. It is not possible to
achieve zero waste discharge, but it is an essential to treat the waste. |
| Among the toxic heavy metal ions which are potential health hazards to
aquatic animals and human like Pb, Cd, Cr, V, Bi and Mn are important. |
| The maximum tolerance limit for vanadium for public water supply are
10 mg/L. Toxicity of metal depends on the type of metal, dose and the ionic
form. Toxicity of vanadium (WHO, 1998) include rhinitis, blood discharge,
an itching and burning sensation in the throat, dry cough with small amount
of viscid sputum, general weakness. The moderate toxicity includes respiratory
tract infection, bronchitic. bronchopneumonia, Disorder of the nervous
system and tremor of the linger and hands. In animals, it affects the kidney,
nervous, respiratory, cordiovaschulai and immune system. |
| Literature survey reveals that, there are many methods namely coagulation,
precipitation,
ions exchange and adsorption, for removal of vanadium metal
ions from aqueous medium. However, adsorption is an easy and economical
process for removal
and retrieval of cation from aqueous medium. Efficiency
of adsorption process
mainly depends on nature of adsorbent, adsorbate, pH.
concentration, temperature,
time of agitation etc. |
| These cheap and efficient absorbents can carry to cater the need of population
in the rural areas and the population in the industrial area where
sate drinking water is not available. In the present study, Vanadium (V5+) is
removed by using coconut shell (Ayub and Iqbal, 2003) as a adsorbent. |
Adsorbent |
| The coconut shell was first dried at a temperature of 160”C for 6 hours. After
grinding
it was sieved to obtain average particle size of 200 mesh. It was then
washed several times with distilled water to remove dust and other impurities.
Finally it was dried again in an oven at 50°C for 6 hours. The adsorbent was
then stored in desiccator for final studied. |
Batch study |
| The dried amount of 0.5 gms of coconut shell was taken in 250 mL reagent
bottle and synthetic solution (200 mL) containing various concentration of
vanadium ion was added and system is equillibrated by shaking the contents of the flasks at room temperature so that adequate time of contact between
adsorbent and final concentration
of metal ion vanadium (V) was determined
by spectrophotometry using hydrogen peroxide method and measured absorbance
at 450 monometer. The spectrophotometer, systronic (model 104)
was used to measure the concentration of vanadium (V) ions. |
RESULTS AND DISCUSSIONS |
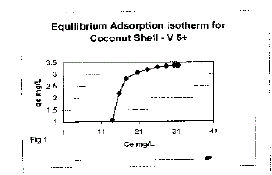
| Equilibrium adsorption isotherm for Ce verses qe plotted for coconut shell
are shown in figure 1. The adsorption capacity in mg/L was calculated then
the equation. |
| qe = (C0 - Ce)V/M where, Co is the initial concentration of vanadium (V)
Ce is the concentration of (V) at equilibrium in mg/L
V is the volume of solution in litre and
M is the mass of adsorbent in grams |
Adsorption isotherms |
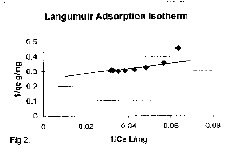
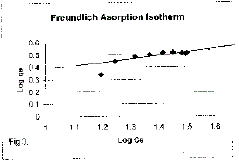
| Equilibrium isotherms was studied for both Langmuir and Freundlich isotherms.
The results are shown in Figure 2 and 3 which, illustrate the plot
of Langmuir and Freundlich isotherms of coconut shell for Vanadium. The
saturated monoluyer can be represented by : |
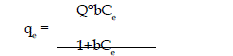 |
| The lineaiised form of the Langmuir isotherms is |
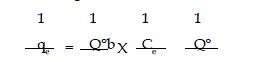 |
| where Q” and b are Langmuir constants. The plot of 1/C, Vs 1/q was found
to be linear, indicating the applicability of Langmuir model. The parametrs Q”
and b have been calculated and presented in Table 1. The Langmuir constant
Q” is a measure of adsorption capacity and b*is the measure of energy of
adsorption. In order to observe
whether the adsorption. In order to observe
whether the adsorption is favourable
or not, a dimentionless parameter ‘R’
obtained from Langmuir isotherm is |
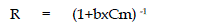 |
| where b is Langmuir constant and Cm is maximum concentration used in
the Langmuir isotherm. The adsorption of Vanadium on Coconut Shell is
a favourable process as “R” values lie between zero to one. Coefficients of
co-relation (r) are also shown in Table 1. The applicability of Freundlich isotherm
was also tried using
the following general equation |
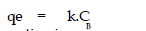 |
| lineaiised form of this equation is |
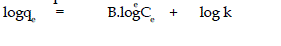 |
| where B & k are Freundlich constants. These constants represent the adsorption
capacity
and the adsorption intensity respectively. |
| Plot of log qe Vs log Ce was also found to be linear. The values of B and
k are presented in Table l. Since the values of B are less than 1, it indicates
favourable adsorption. |
Effect of concentration of metal ion and contact time |
| The responce of Adsorbate dose and contact time on the removal of V( V) is
presented
in Figure (I). The observations reveal that an increase in the adsorbate
dose, rate of adsorption increase upto certain level and then it become
constant. Also as the time of contact increase, adsorption increase and then
it become constants. |
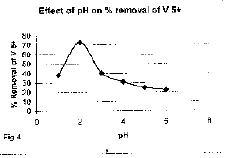
Effect of pH on the removal of vanadium (v) |
| The effect of pH on the removal of vanadium (V) is shown in Figure[4]. Experiment
were conducted at the constant initial vanadium (V) concentration,
adsorbent dose (coconut) of 0.5 gm/100 mL and the contact time of 4 hours.
The pH of the aqueous solution is an important controlling parameter in the
adsorption process. It was observed that the percentage removal of V(V) is
higher at pH = 2 and then decrease with increase of pH. |
Effect of particle size |
| The adsorbent particle size has significant influence on the kinetics of adsorption.
The influence of particle size furnishes important information for
achieving optimum
utilisation of adsorbent. Four particle size 50, 100, 150. 200
micron size (Indian
Standard Sieves) under optimum condition. It is found
that, as the particle size increase the rate of adsorption decrease. |
Kinetics of adsorption |
| 0.5 gm of coconut shell and 200 mL V5+ solution was taken in 1000 mL R.B.
and shake vigorously for about four hours. After every 15 minutes. 5 mL
sample of the solution was withdrawn for the first hour and subsequently the
interval between the samples withdrawn was increased to 30 minutes. The
concentration of the metal ions in the sample, withdrawn were determined
by the spectrophotometi\and were designated as C( and the value of the
concentration of the metal ion on the coconut shell at the same time interval
estimated using the relation. |
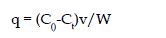 |
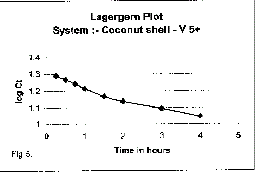
| The rate of adsorption of V5+ on coconut shell was studied by using the
first order rate equation proposed by Lagergren (1998). |
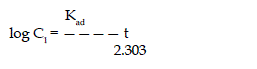 |
| where Kad is the rate constant for adsorption. The plote of log Ct Vs t is
shown in figure (Lagergren, and Bil 1998). |
CONCLUSION |
The following conclusions have been drawn from the presents study |
| 1. The percentage retrieval of vanadium is formed to be increase with
decrease the initial concentration of vanadium. The removal is found
rapid in initial stages followed by slow adsorption upto saturation limit. |
| 2. The developed technique of retrieval of vanadium ions using coconut
shell appears to be a cheap and practically viable for the use of semiskilled
worker in the villages. |
| 3. The present work on adsorption process is in good agreement with
Langmuir isotherm indicating monolayer adsorption process. |
| 4. The result on adsorption process reveals that at pH = 2.0. vanadium
uptake capacity is better. |
| 5. The straight lines plots of logC( Vs t for the adsorption’show the
validity of Lagergren equation and suggest the first order kinetics. |
| 6. Regeneration studies are not necessary with the view that the cost of
the adsorbent is very low and it can be disposed of safely. |
Tables at a glance |
 |
| Table 1 |
|
| |
Figures at a glance |
 |
 |
 |
 |
 |
| Figure 1 |
Figure 2 |
Figure 3 |
Figure 4 |
Figure 5 |
|
| |
References |
- AyubSohail and Iqbal Ali, S. 2003. Chromium removal by adsorption on coconut shell, IAEB. 30 : 30-36. Tomer, Mamta 1999. Quality Assessment of Water and Wastewater. Lewis Publisher Boca Ratan.
- Singh, D.K. and LalJyosna, 1992. Removal of toxic heavy metal ion from waste-water by coal based adsorbent. Pollution Research. 11 : 37-42.
- Environmental Health Criteria 81, 1998. Published by W.H.O.Genewa. 101.
- Lagergren, S. and Bil K. 1998. SvenskaVatenskapsakadhand 24.
|